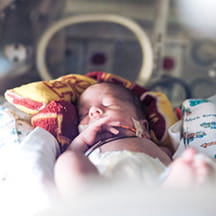
For decades, outcomes for the most severe cases of vein of Galen malformation (VOGM) have remained stubbornly poor, even at the most experienced children’s hospitals.
A study from researchers at Boston Children’s Hospital, published in the Journal of the American Medical Association (JAMA), suggests that in-utero embolization can significantly improve survival and neurodevelopmental outcomes in these highest risk patients.
The study builds on the hospital’s pioneering work in fetal treatment of VOGM, led by Darren Orbach, MD, PhD, chief of neurointerventional radiology and co-director of the Cerebrovascular Surgery and Interventions Center, providing further evidence of its feasibility.
Orbach talked with the Children’s Hospital Association about how the fetal approach differs from standard care, why timing matters, and what the work could mean for the future of pediatric cerebrovascular intervention.
What is significant about this study?
This study showed a real change in outcomes for a group of patients we’ve historically had very little success with. We enrolled seven fetuses with severe VOGM who, based on fetal imaging, were at extremely high risk for postnatal collapse. We were able to successfully perform in utero embolization in five of them. Three of those five survived past the neonatal period, and all three are developing without neurodevelopmental delay.
For this particular risk group, historical mortality has been close to 90%. In this cohort, mortality dropped to 43%.
Why does intervening before birth make a difference?
The advantage isn’t just earlier treatment — it’s the physiologic environment in which the intervention occurs. In utero, the placenta acts as a massive buffer. It can absorb enormous blood flow that would otherwise overwhelm the newborn heart and brain after birth.
At the same time, fetal cerebral circulation is operating at a much lower baseline demand than postnatal circulation. By reducing flow through the malformation under those conditions, we’re taking advantage of a window that simply doesn’t exist after delivery.
Once that window closes, the risks increase dramatically.
How is fetal embolization different from standard postnatal embolization?
Postnatal embolization typically involves accessing arterial feeders through the umbilical artery or the groin and navigating very small, fragile vessels in a critically ill newborn. That approach can be lifesaving, but it is one of the most technically challenging procedures we do, and it carries significant risk, particularly when done under emergency conditions.
In the fetal procedure, we take a completely different route. We access a large venous sinus at the back of the fetal skull and place coils directly into the central vein of the malformation. The goal is not a complete cure before birth but enough flow reduction to stabilize the newborn after delivery and prevent immediate collapse.
It’s still embolization, but the target and the timing are fundamentally different.
What do we know so far about safety?
In this initial cohort, we did not see procedure-related hemorrhage or brain injury, which are real concerns in postnatal embolization of critically ill newborns. That said, this was designed as a feasibility study, not a definitive safety trial.
The reason we’re moving into the next phase is to rigorously answer that question — how safe this is, how reproducible it is, and how consistently it improves outcomes when applied under strict criteria.
Why is patient selection so important?
This procedure is only appropriate for a very specific subgroup of patients. Advanced fetal MRI allows us to identify fetuses who are at extremely high risk for postnatal collapse and poor overall outcomes, but who do not yet have brain injury.
If there’s already brain injury in utero, closing the malformation won’t reverse that damage and may actually increase the risk of hemorrhage. In those cases, intervention is unlikely to help.
That’s why strict inclusion and exclusion criteria are essential, and why this work is being done in a controlled trial setting.
Could most children’s hospitals offer this approach?
To do this safely and effectively, you need deep expertise in fetal surgery and extensive experience managing VOGM across the neonatal period. Even after fetal intervention, many infants still require complex NICU, cardiology, and neurointerventional care after birth.
This is not a stand-alone procedure. It’s part of a highly specialized continuum of care, and it will likely remain concentrated in a limited number of centers.
What does this mean for children with VOGM?
In the near term, it offers a path to improved survival — and potentially better long-term neurological outcomes — for a condition that has long been associated with catastrophic postnatal failure.
More broadly, this is the first fetal brain intervention designed specifically to modify blood flow. If we can show that this approach is safe and effective, it changes how we think about when intervention should occur, not just for VOGM, but potentially for other conditions as well.
For a field that has traditionally focused on crisis management after birth, that shift may be just as important as the outcomes themselves.